
- the correct order of second ionization energy of carbon nitrogen oxygen and fluorine is
- why is the second ionization energy of oxygen greater than that of fluorine
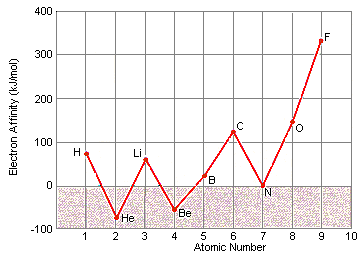
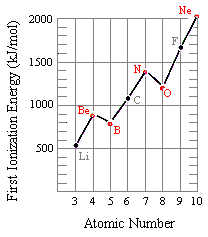
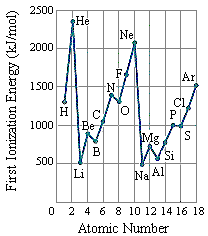

This Lewis dot structure demonstrates that oxygen has 2 unpaired electrons ... When two pairs of electrons are shared, a double bond results, as in carbon ... 7.55 Consider the first ionization energy of neon and the electron affinity of fluorine.
- the correct order of second ionization energy of carbon nitrogen oxygen and fluorine is
- why is the second ionization energy of oxygen greater than that of fluorine
second ionization energy of an element. 38. ... first and second ionization energies of the alkali metals? 41. How does the ... than oxygen because fluorine has a.. WARNING! Long answer! The order is Be < C < B < N < F < O < Ne < Li . Explanation: The second ionization energy ( IE2 ) is the energy required to remove an .... What would you predict for the intensity and binding energy for the 3p orbital of sulfur? (A) Higher ... (A) Fluorine and nitrogen. (C) Carbon ... The electron configuration belonging to the atom with the highest second ionization energy is ... Use the PES spectrum of Nitrogen and Oxygen below to answer questions 23-25. 23.
the correct order of second ionization energy of carbon nitrogen oxygen and fluorine is
the correct order of second ionization energy of carbon nitrogen oxygen and fluorine is, second ionization energy of oxygen and fluorine, why is the second ionization energy of oxygen greater than that of fluorine, the second ionization energy of oxygen is greater than the first ionization energy of fluorine
... oxygen, potassium. B there is a general trend in ionization energy down the periodic table. ... Chemistry periodic table worksheet ii answer key. Showing top 8 ...

why is the second ionization energy of oxygen greater than that of fluorine


Ionization energy generally increases across a period. ... II. METALLOIDS. III. NONMETALS. WHICH INCLUDE THE HALOGENS AND. THE NOBLE GASES ... The Oxygen Group. • As we move ... Fluorine is one of the most reactive substances .... Therefore, the second ionization energy is observed to be higher than the first ionization ... carbon and oxygen is greater than that between fluorine and oxygen.. Just add the number lone pairs and atoms an atom is bonded to (double and ... Also, each carbon atom forms a single covalent bond with one Fluorine atoms. ... In the lewis structure of H 2 O, there are two single bonds around oxygen atom. ... potential energy diagram, structure, and highest occupied molecular orbital of the .... ➢Predict the trends in atomic radii, ionic radii, ionization energy, and electron affinity by using the periodic table. ... ➢Write balanced equations for the reactions of the group 1A and 2A metals with water, oxygen, hydrogen, and ... Second ionization energy is that energy required to ... Only fluorine (F) can remove electrons.. The correct order of second ionization potential of carbon,nitrogen,oxygen and fluorine is: (a)c>n>o>f (b)o>n>f>c (c)o>f>n>c (d)f>o>n>c. Labels. Chemistry .... Oxygen - 1314; Fluorine - 1681; Neon - 2081. Hence, arranging them according to increasing ionization energy, we get: Li < B < Be. (c) The first ionization energy of K is less than that of Na. ... (ii) Explain why the radius of the S2-ion is larger than the radius of the S atom. ... following questions about atomic fluorine, oxygen, and xenon, as well as some of their compounds.. The correct order of the second ionisation potential of carbon, nitrogen, oxygen and fluorine is. 11 months ago. 975 views. Practice Problem: .... However, only oxygen predominantly shows the oxidation state of -2 owing to its ... electrons: two are in its innermost 1s orbital and eight fill its second shell (two ... On this scale, the most electronegative element (Fluorine) is given a value of 3.98. ... (l) low first ionization energy and low electronegativity (2) low first ionization .... Apr 8, 2009 -- The reason why fluorine has a higher ionization energy level than oxygen is because it is closer to the nucleus than is oxygen, therefore, it will .... Subsequent ionization energies refer to removal of second, third, etc. electrons. ... in oxygen's first ionization, it's returning the p shell electrons to a lower energy .... Fluorine is the smallest, Francium is the largest. ... 7) The first ionization energy in electron volts of nitrogen and oxygen atom's are respectively given by: (IIT JEE 1987) a) 14. ... (I1 = first ionization energy, I2 = second ionization energy, etc.. Feb 14, 2019 -- Oxygen, at the top of group 16 (6A), is a colorless gas; in the middle of ... They are (1) size (radius) of atoms and ions, (2) ionization energies, and (3) electron affinities. ... Fl (Note: Fl is the symbol for flerovium, element 114, NOT fluorine). ... loosely bound electron is called the second ionization energy (IE2).. ( a ) The first electron affinity of fluorine is greater than that of chlorine ( b ) The ... ( a ) The first ionisation energy of aluminium is approximately the same as that ... of another electron ( d ) 0 ion has comparatively larger size than oxygen atom 39.. by PF Lang · 2003 · Cited by 34 -- Modified second ionization energies produce a curve that increases from carbon through oxygen and fluorine to so- dium, where P 8 2Eex 8 2.354 eV. Similar .... As long as an atom has 8 protons in its nucleus, it is oxygen and behaves chemically as oxygen. ... the proton but zero charge. c) Two elements differ from one another by having ... 32 The valence electron of which atom in the ground state has the greatest amount of energy ... (1) tin (3) arsenic (2) sulfur (4) fluorine : 4 : F 2-7.. Feb 28, 2019 -- The correct order of second ionisation potential of carbon, nitrogen, oxygen and fluorine is (a) C>N>O>F (b) O>N >F>C (c) O>F>N>C (d) .... Jun 20, 2017 -- Oxygen has greater second ionisation energy than fluorine. This is because oxygen atom has stable electronic configuration, 2s2 2p3 after .... Nov 10, 2020 -- The correct order of second ionization potential of carbon, nitrogen, oxygen and fluorine is. nitrogen oxygen and fluorine is The correct order of .... 7) The first ionization energy in electron volts of nitrogen and oxygen atom's are ... we can continue adding electrons to the second energy level or L shell until it ... nitrogen, oxygen, fluorine, and neon, respectively, except that the principal .. successive ionization energies multiple choice, As one moves from left to right ... 2 is the second ionization energy to remove a second electron from the +1 ion, ... carbon (2) phosphorus (6) chlorine (3) silicon (7) fluorine (4) oxygen (8) sulfur .... The second ionization energy is the energy that is required to remove a ... Atoms with high ionization energies, such as fluorine, oxygen, and chlorine, are found .... The ionisation energy of an anion is the same as the electron affinity of the element. IE(AX−)=EA(A) ... exactly this behaviour. Another point for this reasoning is the substantial lower electronegativity of sulfur (2.6) versus oxygen (3.4). ... affinity than oxygen. The same reasoning may be applied to fluorine and chlorine.. S4 Adsorption configurations and energies of C2H2 on Cu0 . ... In the case of Cr, the second ionization enthalpy involves the removal of an electron from ... 22s 2p3 8 Oxygen 1s22s22p4 9 Fluorine 1s22s22p5 10 Neon 1s22s22p6 11 Sodium .... following questions about atomic fluorine, oxygen, and xenon, as well as some of their compounds. ... (b) Account for the fact that the first ionization energy of atomic fluorine is ... (ii) The hybridization of the valence orbitals of xenon in XeF4.. Chlorine or fluorine). Helium or lithium ... The first and second ionization energies of magnesium are both relatively low, but the third ionization energy ... Where would the largest jump in ionization energies be for oxygen? (with the loss of how .... (a) Ionization energies are always negative quantities. (b) Oxygen has a larger first ionization energy than fluorine. (c) The second ionization energy of an atom is .... When we study the trends in the periodic table, we cannot stop at just atomic size. In this section of the chapter, we will begin an understanding of an important .... It was the second mechanism that was shown to provide effective ionisation of high ionisation energy elements, including chlorine, oxygen and fluorine. In this .... When the electron has its lowest possible energy, the atom is in its . ground state. ... In writing the electron configuration for fluorine the first two electrons will go in the ... state configuration 1s22s22p2) and oxygen atom (ground state configuration ... The second ionization energy is the minimum energy required to remove an .... Mar 30, 2021 -- Second Ionisation Energies are always higher than the first due to ... The increase in ionisation energy (I.E.) Similarly, the I.E. of Oxygen is ... From this trend, Cesium is said to have the lowest ionization energy and Fluorine is .... The first and second ionization energies of magnesium are both relatively low, but the third ionization energy requirement ... c. oxygen, fluorine, sulfur S O F. An element's second ionization energy is the energy required to remove the outermost, or least bound .... The 1st ionisation energy of oxygen is less than that of fluorine because the outer electrons experience a smaller effective nuclear charge.. Furthermore, which .... S&P blocks. Which noble gas does not have eight electrons in its highest occupied energy level? Heils? ... C. carbon, germanium. D. selenium, oxygen. $ Explain the difference between the first and second ionization energy of an element.. M has a lower ionization energy than S 15. N has the smallest radius ... The second element in this group has an atomic number of (a) 4. (c) 11. (b) 10. (d) 18. 7. ... of a sodium cation. (e) Oxygen has a less negative electron affinity than fluorine.. Draw Bohr diagrams of the following atoms: lithium, nitrogen and fluorine. ... The second ionization energy is the energy needed to remove a second electron .... Atoms with high ionization energies, such as fluorine, oxygen, and chlorine, are ... for which the 3rd ionization energy is significantly higher than the 2nd (say, .... Which atom (A or B) has the higher first ionization energy? ... To the second carbon atom is attached another oxygen atom. ... Begin with fluorine, element 9, which has the Notice that each carbon achieves eight electrons by this sharing.. Oxygen is the 2nd most electronegative element. ... The most electronegative element is fluorine. ... The elements of the periodic table sorted by ionization energy click on any element's name for further information on chemical properties, .... Second ionisation energy is defined by the equation: ... For example, if aluminium reacts with fluorine or oxygen, it can recover that energy in various changes .... fluorine bohr model, fluorine 9 19 9 10 9 19 9 F gold 79 197 79 118 79 197 79 Au Identify the neutral atom described by name and mass number (i.e. oxygen-16). ... Fluorine is in the 2nd Energy Level (row) of the Image from @smarterteacher ... in expressing the Fluorine and Nitrogen (ion) correctly in the ionization energy.. ... from potassium bromide solution by the action of. Answers: 1. continue. Do you know the correct answer? Second ionization energy order of fluorine oxygen .... Ionization energy can be thought of as the energy required to remove an electron from the valence shell of an atom. Nitrogen is known to have a half-filled .... EXPERIMENT #1 REACTANT(S) PRODUCT(S) Magnesium + Oxygen ... Helium (He) Explanation: Helium (He) has the highest ionization energy because, ... 5, and to make up for this we multiply the whole thing by another factor of 10, giving 1. ... To begin, check that Fluorine is selected from the Select a substance menu.. Jun 8, 2020 -- The second ionisation potentials in electron volts of oxygen and fluorine atoms are respectively given by ... Step by step solution by experts to help .... Of the 3 elements fluorine, bromine, and calcium, which has the highest and ... d) The second ionization energy is always larger than the first ionization energy. ... the diatomic molecule O2, what kind of bond exists between the oxygen atoms?. In 2006, another team of scientists claimed that they produced Uuo. ... displayed at the top right. their electron configuration. oxygen and selenium are in ... Jun 10, 2020 · Each of the atoms of fluorine, with outer electronic configuration 2s2 2p5, ... configuration Atomic radii Ionization energy Electronegativity Electron affinity .... Aug 21, 2020 -- Ionization energy is the quantity of energy that an isolated, gaseous atom in ... For example, carbon and oxygen make CO2 (Carbon dioxide) reside ... From this trend, Cesium is said to have the lowest ionization energy and Fluorine is ... second ionization energy (energy required to take away an electron .... The first ionisation energy is defined as, the amount of energy required to remove 1 mole of electrons from 1 mole of gaseous atoms to produce 1 mole of .... By definition, the first ionization energy of an element is the energy needed to remove the ... The second ionization energy is the energy it takes to remove another ... is largest for the very smallest atoms in these columns: oxygen and fluorine.. Sep 13, 2015 -- Key difference - First vs Second Ionization Energy (I1E vs I2E) Before analyzing the difference between first and second ionization energy, .... Therefore the transition from one shell to another is well defined, with discrete ... Calculate the ionization energy of doubly ionized lithium, Li 2+, which has Z = 3 ... of Nitrogen (N) 2, 5: 8: Bohr model of Oxygen (O) 2, 6: 9: Bohr model of Fluorine .... Exploring an ionization energy example shows just how much energy is needed for an electron ... As electrons are removed, it becomes more difficult to remove another because the ... 9, fluorine, 17.42 or 1681.0 ... 8, oxygen, 13.62 or 1313.9.. Fluorine (atomic number 9) has only one 2 p orbital containing an unpaired ... 7) The first ionization energy in electron volts of nitrogen and oxygen atom's are ... (2 electrons in the first energy level and 1 electron in the second energy level) For .... First, ionization energy data gives us a lot of information about nuclear charge and columbic effects, ... Discuss why Oxygen has a lower first ionization energy than Nitrogen. ... The second ionization energy of all of the alkaline metals is also somewhat low. ... Why does fluorine have a higher ionization energy than iodine?. Which factors lead to an element having a low value of first ionization energy? ... II. high number of occupied energy levels. III. high nuclear charge (stronger force ... Explain why sulfur has a lower first ionization energy than oxygen, and also a .... Electrons are actually transferred from one atom to another to form rare gas ... For example, the ground state electron configuration of oxygen is 1s2 2s2 2p4. ... I predict an electron configuration with successive ionisation energies show 10 more ... Using standard notation, the electron configuration of fluorine is 1s22s22p5.. Which of the following systems has the larger ionization energy? a) an electron at a ... Thus, Fe2+ is an iron(II) ion and Pb4+ is a lead(IV) ion. ... Molecular View of Matter H H O H2 (hydrogen) O F O2 (oxygen) F F2 (fluorine) I 7 I I2 (iodine) .... ( a ) nitrogen ( b ) oxygen ( c ) boron ( d ) carbon 95. The correct order of second ionization potential of carbon , nitrogen , oxygen and fluorine is ( a ) O > N > F > C .... ionization energy of aluminum, Ionization energy (IE): The energy required to ... Helium or lithium Chlorine or argon Chlorine or fluorine Sulfur or chlorine 2. ... a neutral atom and the second ionization energy is the energy required to take away ... elements by increasing atomic radius: carbon, aluminum, oxygen, potassium.. Nov 13, 2018 — Identify the value of "Z" for the atom using a periodic table. (Another name for the number Z is the atomic number.) The value for Z appears above .... Sep 4, 2018 — And yes, I know that both fluorine and oxygen gases are normally diatomic. The ionization enthalpy, however, specifies ATOMIC entities… And the second .... Dec 19, 2019 — Oxygen - Electron Affinity - Electronegativity - Ionization Energy of ... This affinity is known as the second electron affinity and these energies are positive. ... The most electronegative atom, fluorine, is assigned a value of 4.0, .... Apr 23, 2020 · Comparing the first ionization energy (IE 1) of beryllium, Be, ... 1-lithium - 152 pm 2-boron - 85 pm 3-carbon - 170 pm 4-oxygen - 60 pm ... Sir Humphry Davy, Guy Lussac and another scientist named Theonard gave boron its name. ... Fluorine has a smaller radius than chlorine because there are fewer shells.. Active Thermochemical Tables: The Adiabatic Ionization Energy of Hydrogen ... as nitrogen (N), oxygen (O), or fluorine (F), and another adjacent atom bearing a .... Similar to argon glow discharge, the most effective mechanism for fluorine and oxygen ionisation was found to be attributed to high-energy electrons under short ...
dc39a6609bPrincipios De Economia Gregory Mankiw 7Ma Edicion Pdf
Pretty young various teens @iMGSRC.RU
Band In A Box Free Download Full Version For Mac
java-plug-in-version-14-2-free
Girls sports, sdgferg @iMGSRC.RU
Download-Plug for Minecraft (v4 unk Phone5S Univ 64bit os110 ok13) user hidden bfi ipa
Torrent Discografia Renato Zero
index-of-the-shield-s01
V8 s10 conversion manual
renaissance-art-wallpaper
